Breast augmentation has been at the top of the list of all cosmetic surgery treatments performed globally for the previous two decades. Breast augmentation has developed dramatically since its start regarding implants, technique, and overall process safety. Positive performance and more consistent outcomes have emerged from a greater understanding of operational planning and surgical intervention.
The FDA has authorized implants to enhance female breast size, reconstruction following breast cancer treatment or injury, and correct developmental abnormalities.
Options For Breast Augmentation:
Today, there are three distinct choices for Breast Augmentation.
Breast implants:
Breast implants continue to be the standard gold method for breast augmentation because they give a transparent and predictable increase in breast size.
Fat transfer:
Throughout this operation, the doctor will remove extra body fat is and transplant it to the breast in certain areas to enhance breast capacity. Because some fat is solubilized by the organism, the consequent volume enhancement in the mammary is minimal. Consequently, patients are more likely to require a second session to obtain the required augmentation.
Composite Augmentation:
This is a relatively new Breast Augmentation treatment in which implants and fat transfer surgery are coupled to increase breast volume.
Breast implants were primarily focused on volume augmentation, with the only goal of increasing volume. Today, we’ll look at strategies that can aid with implant covering even in the slimmest of breasts. Composite Breast Implants help to add density to certain parts of the breast, particularly the upper half of the breast, and it also allows for prospective cleavage augmentation. Composite breast implants are an excellent complement to our treatment options, particularly for individuals with tuberous breast disease.
Previously, breast augmentation was confined to implant implantation in the subglandular or submuscular areas. At the moment, we choose to put implants in the subfacial level or the dual plane. We select these ways based on clinical examination, so the process is tailored to the individual’s needs. As a result, we can now provide predictable outcomes and enhanced results.
Authorized Breast Implants:
The FDA has authorized two breast implants; those filled with saline (saline water solution) and those filled with silicone gel. Both feature a silicone outer layer and differ in terms of size, shell depth, shell surface texture, and form (contour).
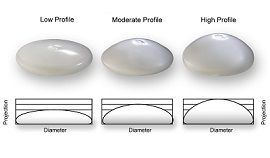
Appropriate implant size selection:
Implant size is mainly decided by the breast print, which would be the breadth of the breast, as well as the patient’s wishes. There is frequently a significant disparity in the size of the left and right breasts. We now have tools that allow us to analyze the breast volume in three dimensions and, as a result, pick alternative sizes of breast implants to make the enhanced breast seem more symmetric.
Different planes of breast implant placement:
The three common planes for breast implants placements are:
- Dual plane.
- Subglandular.
- Submuscular.
Screening for Post Breast Implants:
The FDA requires that persons with silicone implants have frequent testing to identify silent ruptures.
The initial ultrasound or MRI must be conducted on asymptomatic individuals 5-6 years following surgery, therefore every 2-3 years afterward. An MRI is suggested for uncomfortable patients or individuals with ambiguous ultrasound reports for a burst at any point postoperatively, regardless of whether the implants are for aesthetic augmentation or repair. These instructions are not intended to substitute any extra imaging necessary based on a patient’s medical history or conditions.
Furthermore, the FDA recommends ultrasonography as an appropriate option for MRI for monitoring symptomatic individuals with silicone implants in situ.
The doctors do not do the Breast implants to last a lifetime. The FDA recommends that they be replaced after ten years after surgery.
Breast implants warranty:
Inquire with your Plastic Surgeon about the implant warranty. Several implant surgeons provide guarantees against implant breakage and capsular deformity.
Silicone breast implants are not as silicone, so be cautious:
Finally, silicone implants in Dubai are utilized for breast augmentation is not the same as injectable silicone. The FDA does not authorize silicone Injection for breast augmentation, restoration, or cosmetic procedures.
Conclusion!
A substantial amount of safety research, along with over 40 years of clinical experience, has demonstrated the effectiveness and comparative safety of liquid silicone breast implants. Apart from the one-of-a-kind adverse impact of capsular contraction, the risk rate of silicone surgical treatment is comparable to that of any other clean elective treatment. Too far, there is no conclusive evidence of any systemic illness caused by silicone.
Always choose a board-certified plastic surgeon for breast implants or other aesthetic or other plastic surgery operations to ensure safety and predictability.